Potential Energy Internuclear Distance Graph
You lot may be thinking, what are intramolecular forces?
Intramolecular forces are the ones betwixt 2 atoms in a molecule! This is very dissimilar from intermolecular forces, which we learn in unit three.
These two are confused often😕, but hither are some tips💡:
-
Intermolecular are between molecules. Recollect inter = between two groups
-
Intramolecular are between 2 groups in a molecule, so remember intra = inside a group.
So far nosotros learned well-nigh two intramolecular forces: covalent bonds and ionic bonds.
Types of Covalent Bonds
You must think the following key data for the AP exam.
Concrete or chemical processes can be described through energy diagrams. In molecules, bonds can either be classified as unmarried, double, or triple bonds.
Unmarried = two electrons involved
DOUBLE = four electrons involved
TRIPLE = 6 electrons involved
An easy way to retrieve this is that each nuance on a lewis dot diagram corresponds to two shared electrons.
Image Courtesy of Shodor
As well notation that:
-
Unmarried bonds are the longest in length and have the smallest bail energy
-
TRIPLE bonds are the shortest in length and take the largest bond energy
Allow's see this information in some graphics👀!
Bond Length and Bond Energy
For covalent bonds, bail length is influenced by the bond order (single, double, triple) and the balance between repulsive and attractive forces. Bond length is the physical altitude betwixt ii atoms bonded to one some other. Bail free energy in the diagram shows how the greatest potential energy is the repulsion of two atoms.

Image Courtesy of SplainScience
Let'due south take a wait at each of these stages:
-
Repulsion - Since the atoms are very shut together and the internuclear distance is very modest, the atoms are experiencing lots of electron-electron repulsion. This is very unstable and leads to a potential energy of greater than 0.
-
Some overlap/attraction - This is the nigh stable state. There is a residuum⚖️ between the repulsive and bonny forces and a stable bond is formed. This has been went over before only hopefully now yous empathise why potential free energy is lowest when the bail is stable.
-
The potential energy at this stage is the amount required to break the bail.
-
-
No overlap/allure - Since the internuclear distance is and so large, in that location is no bond formed. This leads to a potential energy of almost 0.
Example
It is skilful to empathise these backdrop considering you may be asked to gauge where an element falls on this graph.
Say the following image is a diagram of chlorine atoms bonded together (Cl-Cl), where would Br-Br autumn in comparing to chlorine'south curve?
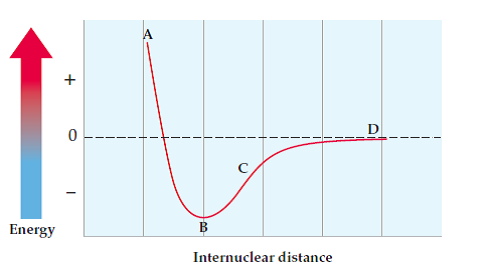
Image Courtesy of Chegg
To answer this question, we have to think about periodic trends:
-
Internuclear altitude: is the Cl-Cl bail or Br-Br bond longer? Well, the one with the largest atomic radii would have to exist the one with the longer bail. As y'all go downwardly in a grouping, atomic radius increases. Therefore, Br-Br is longer than Cl-Cl.
-
This helps out with cartoon the curve in relation to the ten centrality.
-
-
Potential energy: which bail would be easier to break? Cl-Cl or Br-Br? This should automatically make you think of ionization energy. The lower the ionization energy, the easier it would be to suspension the bond --> the less energy needed. Equally you go down a grouping, ionization energy decreases because there are more occupied electron shells. Therefore, Br-Br has a lower ionization energy and this bond is much easier to break.
-
This helps out with drawing the curve in relation to the y axis.
-
Knowing that Br-Br is longer and easier to interruption, you would have to graph its curve up (less energy) and to the right more (larger internuclear distance).
This question is a very good way to test your knowledge nigh this central topic and periodic trends. Here is what the graph should wait like:
Forces Within Ionic Bonds
Understanding the strength of ionic interactions involves the use of Coulomb's police force:
Energy of two interacting charged particles = (Q1*Q2)/d Where charges, Q1 and Q2, are separated past a distance, d
*Attraction occurs if the charges are opposite and repulsion occurs if the charges are the same*
-
Interaction strength INCREASES equally distance DECREASES since those ions tin can exist closer together, so smaller ions have stronger interactions
-
Larger charges on the interacting ions INCREASES interactions since interaction forcefulness is proportional to charge
Coulomb'due south Law is everywhere!
Potential Energy Internuclear Distance Graph,
Source: https://library.fiveable.me/ap-chem/unit-2/intramolecular-force-potential-energy/study-guide/2ybBItwMrCkB9MAOO9lC
Posted by: ricketsonsmagre1960.blogspot.com


0 Response to "Potential Energy Internuclear Distance Graph"
Post a Comment